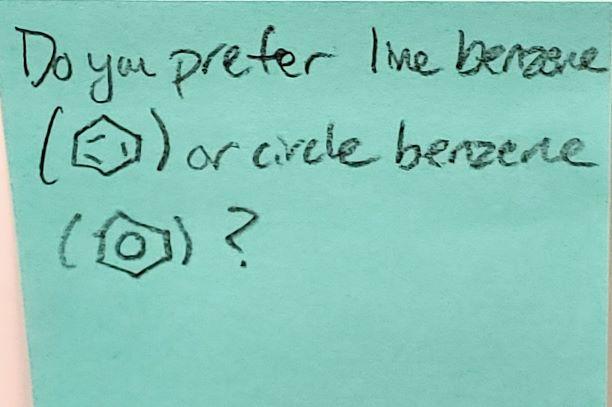
The circle may give a better sense of what the electrons are actually doing, but the lines may help when you’re trying to relate specific things going on with the bonds. Both are generally recognized and understood.

Answering questions at Boston College O’Neill Library
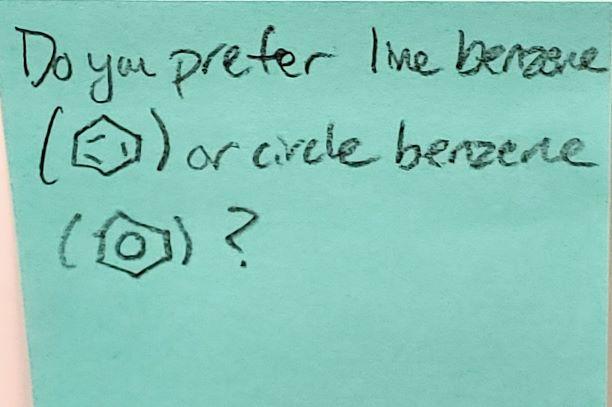
The circle may give a better sense of what the electrons are actually doing, but the lines may help when you’re trying to relate specific things going on with the bonds. Both are generally recognized and understood.

The uncertainty principle states that the position of an object cannot be known simultaneously with its momentum. The more precisely one quantity can be determined, the less precisely the other is known. In terms of helping you study, yes we can. I am certain of that (see what I did there?… I am “certain” of that… got it?…) You can contact the Connors Family Center, located at O’Neill’s second floor. They offer tutoring assistance in general and organic Chemistry. bit.ly/BC-connors

My librarian friends who are blessed with lungs say take a deep breath — it’s OK to chill and regroup. I know you’ve heard it before, but life is a marathon, not a sprint. You may want try chemistry again next semester, or, perhaps, it’s time to redirect your academic focus. Consider checking in with your academic advisor. Take a look at the classic, What Color is your Parachute (O’Neill Library HF5383 .B56). Even though it’s for job hunting/career stuff, it may help you think about what gives you joy, your interests, and your strengths.
I’m afraid the answer is study – but maybe you can study more effectively, if you don’t have the time to study for more hours. I’d check in with both the professor and the Connors Family Learning Center (bit.ly/BC-connors) for advice and tutoring.
![Orgo is so hard ',( [drawings of molecules] [response: same] [response: [in Chinese]]](https://library.bc.edu/answerwall/wp-content/uploads/2019/09/AW09252019-06.jpg)
It’s generally acknowledged to be one of the more challenging subjects. The labs can be fun, though! Ask for peer tutoring at the Connors Family Learning Center (bit.ly/BC-connors) and/or form a study group with your classmates. You can do this. (And, oh, yeah – I totally appreciated the orgo pun in Chinese. We could bond over that. Covalently.)

It’s challenging material!. See if you can get peer tutoring at the CFLC (by May 2, bit.ly/BC-connors) and schedule a meeting with your professor to ask what your clearest path to understanding and passing might be. Touch base with your academic advisor, as well. Take good care of yourself (eat and sleep!) so you have the best possible chance for success. I wish you all the best, but also want you to know that failing a course is not the end of the world, even though it might feel that way at the time. There is always a path forward.
![B/c you are a biological failure :) [Response: mean!! :(]](https://library.bc.edu/answerwall/wp-content/uploads/2019/04/aw043019-10.jpg)
I’m sure they meant well? In the meantime, here’s a joke to lighten the mood: A physicist, a biologist, and a chemist were going to the ocean for the first time. The physicist saw the ocean and was fascinated by the waves. He said he wanted to do some research on the fluid dynamics of the waves and strode into the ocean. Obviously he drowned and never returned. The biologist said he wanted to do research on the flora and fauna inside the ocean and he also strode right in. He, too, never returned. The chemist waited for several hours and then wrote the observation, “The physicist and the biologist are soluble in ocean water.”

Practice, practice, practice! Set the bar high as you study; in a sense, give yourself the chem test over and over before the actual test. And get help, which you can find in the Connors Family Learning Center in the form of tutors: bit.ly/BC-connors

Far from being proof of idiocy, your persistence in the face of adversity is admirable and scholarly. Perhaps you are taking chemistry for a major or pre-professional requirement. Maybe you’re just trying to prove something to yourself. In any case, I wish you the best with it. Do visit the CFLC (bit.ly/BC-connors) for peer tutoring whenever you need assistance.

I’m sure you’re wittig (:P) enough to do the work on this ylide formation on your own, but if you need resources, here’s a handy reference: bit.ly/wittig-reaction. (BTW, it’s “PPh3” not “PhP3”). According to a chemist friend, it might help to think of it as a war: Electrons are the ammo. Some atoms have extra, and we call those lone pairs. Phosphorus is one of these. Some atoms are vulnerable, like the carbon atom that the X is attached to. The lone pair on the phosphorus attacks the carbon, kicks off the X (it’s usually chloride), and now it’s attached to that carbon. The X is now floating around naked and afraid, so it grabs a hydrogen off that carbon it just left and now it’s happy. This creates a double bond between the carbon and the phosphorus, which is the ylide, and which will now go on to do the Wittig reaction.

There are people who hate General Chemistry, but enjoy Organic; very different experiences. The labs can be quite exciting. I won’t kid you; it’s a very tough course. You’d be in there with all the premeds, so the tension level can get intense. I’d talk it through with your Academic Advisor. Maybe you’d thrive on the challenge; as the Organic Chemist said, “It takes alkynes…”

(Wolfman Chem…SOS)
It’s hard! When work slips out from under you, the first question to ask is “why”: is it the difficulty (difficult concepts or processes) or the volume? (Or… argh! both) If difficulty, arrange a tutor through the CFLC: bit.ly/BC-connors. Tutors are really good at breaking complex problems into easier-to-digest component parts. If volume, a) plan out your study times, carefully allotting how much time it *actually* takes (not how much time you wish it would take), b) arrange a tutor. Tutors are good at identifying ways to be more efficient. And go easy on yourself if you can’t finish absolutely everything. It happens.